Food Traceability: Regulations, Systems and Best Practices
As a food manufacturer, you need to demonstrate at any point where your raw materials came from and which customers received your products. This is not optional advice — it is a legal obligation that applies to every link in the food chain, from raw material producers to retailers.
Traceability may sound like an administrative burden, but it is first and foremost a safety instrument. During a recall, you need to determine within hours which batches are affected, which raw materials they contain, and where those products were delivered. The faster you can do this, the smaller the damage.
What Is Food Traceability?
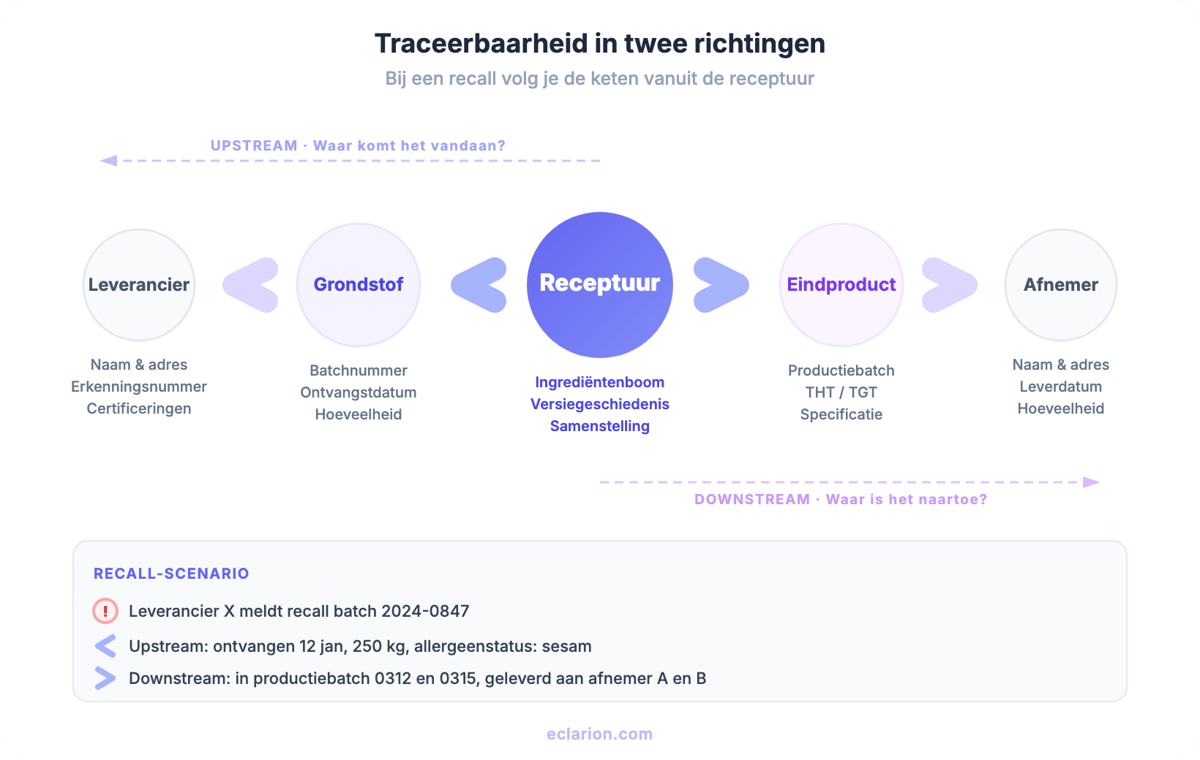
Traceability is the ability to follow a food, feed or ingredient through all stages of production, processing and distribution. It works in two directions:
- Upstream (backward): from finished product back to the raw materials and suppliers
- Downstream (forward): from raw material or product to the customers and ultimately the consumer
European legislation follows the principle of one step back, one step forward. You do not need to oversee the entire chain, but you must be able to identify the direct link before and after you: who did you receive it from, and who did you deliver it to.
Legal Framework: the General Food Law
The foundation for traceability in the EU is Regulation (EC) 178/2002, the General Food Law. Article 18 requires all food business operators to:
- Be able to identify who supplied a food, feed or ingredient
- Be able to identify which businesses they supplied their products to
- Have systems and procedures in place to make this information available to the competent authorities
This applies to every link in the chain. The obligation is technology-neutral: the law does not prescribe how you do it, only that you must be able to.
What Do You Need to Trace at a Minimum?
For each raw material and each product, you need to be able to retrieve the following information:
| Information | Upstream (purchasing) | Downstream (sales) |
|---|---|---|
| Name and address of supplier/customer | Required | Required |
| Product/raw material identification | Required | Required |
| Date of transaction | Required | Required |
| Quantity/volume | Required | Required |
| Batch/lot number | Recommended* | Recommended* |
* For products of animal origin, batch or lot numbers are mandatory under Implementing Regulation (EU) 931/2011, Article 3(g).
The basic obligation under the General Food Law is administrative: who supplied what, when and how much. Batch numbers are not strictly required under Regulation 178/2002, but for products of animal origin there is an additional obligation via Regulation 931/2011. And even without a legal requirement, batch numbers are indispensable in practice: without batch linking, you cannot determine which specific production batches are affected during a recall and would have to recall everything.
The Three Levels of Traceability
Traceability is not binary. There are three levels, and where you stand determines how precisely you can respond during a recall.
Level 1: Supplier Traceability
You know which supplier delivers which raw material. If a problem arises, you can determine which products contain that raw material. But you do not know which specific batch is involved. This is the legal minimum under Regulation 178/2002, but during a recall you end up recalling more than necessary.
Level 2: Batch Traceability
You link specific incoming batches to specific production batches. Supplier X recalls batch 2024-0847. You look it up: that batch was used in your production batches 0312 and 0315. Only those two need to be recalled. This is the level that certification standards such as FSSC 22000, BRC and IFS expect.
Level 3: Internal Process Traceability
In addition to the raw material-to-finished product link, you document the production process itself: who performed which step, at what temperature, with what result. A complete batch record that shows not only what went in, but also how it was produced.
Most food manufacturers operate at level 2. Level 3 is primarily relevant for producers of perishable goods, products with use-by dates, and producers working to pharmaceutical standards.
Traceability and HACCP
Traceability and HACCP are inseparable. The seventh HACCP principle (documentation and record-keeping) requires you to maintain a complete file of your food safety activities. Traceability is a core part of that.
The connection works in both directions:
- From HACCP to traceability: your hazard analysis determines which critical control points you monitor. The records at those control points (temperature measurements, pH values, visual inspections) together with your batch administration form the evidence that a specific production run was produced safely.
- From traceability to HACCP: if a raw material is recalled, you need to use your traceability system to determine which production batches are affected. Then you use your HACCP documentation to assess whether those batches are safe or need to be recalled.
During a food safety authority inspection, traceability is tested as standard. The inspector picks a random finished product and asks you to trace the raw materials, suppliers and production data. You need to be able to do this within a reasonable time. "I'll look it up and email it later" is not an acceptable answer.
What Do You Need to Document?
Traceability stands or falls with three types of records. Together, they form the chain you need to be able to walk through in minutes during a recall.
Raw Material Registration (Purchasing)
For each incoming raw material, you record:
- Supplier (name, address, approval number where applicable)
- Product description and article code
- Batch or lot number from the supplier
- Date of receipt and quantity
- Any deviations on receipt
Production Registration (Processing)
The link where purchasing and delivery come together. For each production batch, you record:
- Production date and batch number
- Raw materials used with their batch numbers
- Quantities per raw material
- Process parameters (temperature, time, pH where relevant)
- Best-before or use-by date
- Any deviations during production
Outgoing Deliveries (Sales)
For each delivery, you record:
- Customer (name, address)
- Product description and batch number
- Delivery date and quantity
- Transport conditions (temperature for chilled/frozen)
Retention Period for Traceability Records
The General Food Law does not prescribe a specific retention period, but the common guideline is: keep traceability records for at least the shelf life of the product plus six months. For products with a long shelf life (canned goods, frozen products), this can run to several years.
Traceability in Your Product Specification
Your product specification forms the foundation of your traceability system. In the specification, you record which raw materials go into the product, which suppliers they come from, and under which conditions the product is produced and stored.
The link between specification and traceability is direct:
- Recipe structure: the recipe defines which raw materials and semi-finished products go into the finished product. This is the structural basis for upstream traceability.
- Supplier linking: for each raw material, you register the supplier including contact details and any certifications.
- EAN/GTIN codes: product identification via barcodes makes automated traceability possible throughout the chain.
- Version control: when the composition changes, you need to demonstrate which version was valid at which point in time. Without version control, your traceability is incomplete.
In Eclarion, you record this information per product as part of your specification file. You manage suppliers centrally and link them to your raw materials. The recipe structure makes the relationship between raw materials and finished products transparent. And version control with immutable snapshots ensures you can demonstrate at any audit what the composition was at a specific moment — including who made the change and when.
From Paper and Excel to a Digital System
Many manufacturers still manage their traceability on paper or in spreadsheets. Purchase receipts in a binder, production forms on a clipboard, delivery overviews in Excel. It works, as long as nothing goes wrong.
But when something does go wrong, speed matters. During a recall, you have hours, not days. And that is when you notice the binder with purchase receipts is not sorted by batch number. That last month's Excel file has a different column layout. That the production registration from shift B is on a different form than shift A's.
Why Excel Falls Short for Traceability
The limitations are the same as with recipe management in Excel: no relationships between data, no audit trail, no structured search. You cannot query with a single click "which finished products contain raw material X from supplier Y, batch Z?" That is exactly the question you need to answer within minutes during a recall.
Why an ERP Module Often Falls Short
ERP systems typically offer batch and lot registration, but the link to product specifications, recipes and labelling is missing. You have your batch administration in the ERP, your recipes in Excel, your specifications in Word and your labels in a separate package. The coherence is missing. And it is precisely that coherence that makes traceability effective: from label, via specification and recipe, back to the raw material and supplier.
Traceability and Certification
Certification standards such as FSSC 22000, BRC and IFS set additional requirements on top of the General Food Law.
FSSC 22000 / ISO 22000
Requires a documented traceability system that is tested at least annually via a traceability exercise (mock recall). The standard does not prescribe a specific timeframe, but in practice auditors apply a guideline of four hours for a complete trace.
BRC Global Standard (BRCGS)
The only major standard that explicitly states the four-hour requirement (clause 3.9). Requires traceability at batch level, an annual mock recall and testing in both directions: upstream (back to raw materials) and downstream (forward to customers). Results must be documented with improvement points.
IFS Food
Requires traceability at batch level and an annual exercise, but adds an extra dimension: the mass balance. The quantities in your traceability system must match your stock administration. This means you can not only demonstrate where your raw materials went, but also that the numbers add up.
For all these standards: the system is assessed on effectiveness, not technology. Whether you do it on paper or digitally does not matter, as long as you can execute it quickly and reliably. In practice, paper-based systems rarely meet the four-hour guideline.
Common Mistakes in Traceability
No Batch Linking Between Purchasing and Production
The General Food Law formally requires only supplier and customer identification. But without batch linking, you cannot determine which production batches are affected during a raw material recall. The consequence: you have to recall all products containing that raw material, regardless of the batch. That is expensive and unnecessary.
Traceability on Paper Only
Paper-based systems are not tested until there is a problem. And then it turns out that forms are missing, illegible, or not filed in the right place. Digital systems enforce structure: you cannot close a production batch without filling in the required fields.
No Periodic Testing (Mock Recall)
If you do not regularly test your traceability system, you do not know if it works. A mock recall takes a few hours and always yields improvement points. Do it at least annually, and after every significant change to your system or processes.
Missing Version Control
Your recipe changes. Your specification gets updated. Your supplier changes. If you cannot demonstrate which version of your recipe and specification was valid at the time of production, your traceability is incomplete. This is one of the most common shortcomings found during audits.
No Coherence Between Systems
Batch administration in the ERP, recipes in Excel, specifications in Word, supplier data in a folder. The information exists, but the connections are missing. During a recall, you have to manually puzzle together the complete picture. That costs time you do not have.
Frequently Asked Questions About Traceability
Is Traceability a Legal Requirement?
Yes. Regulation (EC) 178/2002 (the General Food Law) requires all food business operators to have a traceability system. This applies to producers, processors, distributors, transporters and retailers.
How Quickly Do I Need to Be Able to Trace?
The law does not specify a timeframe, but food safety authorities expect traceability data to be available "without delay." Certification standards such as FSSC 22000 and BRC use a guideline of four hours for a complete trace. In practice, customers increasingly expect it within one hour.
Do I Need to Register Batch Numbers?
The General Food Law does not explicitly require this, but certification standards such as FSSC 22000, BRC and IFS expect traceability at batch level. In practice, batch registration is indispensable for an effective recall. Without batch linking, you have to recall everything rather than just the affected batches.
What Is a Mock Recall?
A mock recall is an exercise in which you simulate that a product needs to be recalled. You select a random finished product and trace it in both directions: back to the raw materials (upstream) and forward to the customers (downstream). The goal is to test whether your system works and how long it takes. Certification standards require this at least annually.
How Does Traceability Relate to the Identification Mark?
The identification mark (the oval health mark) is part of the traceability system for products of animal origin. The mark identifies the last processing establishment in the chain and makes it possible to trace the product to a specific business. For products of animal origin, the identification mark is mandatory alongside the general traceability requirements.
How Do I Set Up Traceability as a Small Business?
Start with the basics: register the supplier and batch number for each raw material, and register which raw materials (with batch numbers) you used for each production batch. Register the customer and batch number for each delivery. With those three registrations, you have a working traceability system. Use software that creates these links automatically, so you do not have to search manually during a recall.
Where Do I Record Traceability Data?
In your product specification, you record the structural information: which raw materials, which suppliers, which composition. In your production administration, you record the operational information: which batches, which dates, which quantities. With Eclarion, you manage the structural side: suppliers linked to raw materials, recipe structures that make the raw material-to-finished product relationship transparent, and version control that logs every change with a complete audit trail.
The Common Thread Through Your Quality System
Traceability is not a standalone administrative exercise. It is the backbone of your food safety system. It connects your HACCP plan with your product specifications, your recipe management with your supplier administration, and your labelling information with the actual composition of your product.
The choice is not whether to set it up, but how. Paper and spreadsheets work until there is a recall. An ERP module gives you batch registration but no coherence with your specifications and recipes. A dedicated tool gives you both.
With Eclarion, you manage the structural foundation of your traceability: suppliers, raw materials, recipe structures, product specifications and version history. Everything in one place, everything with a complete audit trail, everything instantly searchable. Start with a free trial and bring structure to your product documentation.